PAIRING THE POWER OF STEM CELLS WITH THE INNOVATION OF ENGINEERING
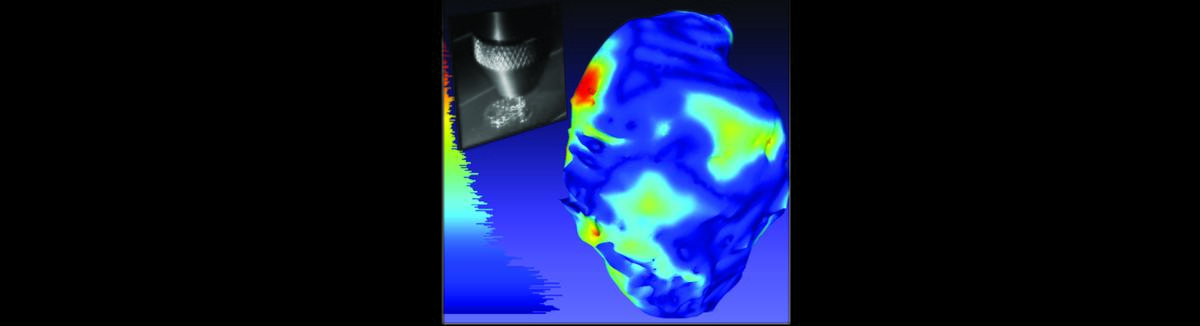
We research mechanisms by which stem cells contribute to tissues during development and we use these insights together with engineering principles to replace damaged systems (especially the cardiovascular system). We are particularly intrigued by spontaneous fusion of stem cells with mature cell types including cardiomyocytes. We have found that cardiac programming can occur in this context and are currently probing the transcriptional profile of stem cell-cardiomyocyte fusion products. We are also interested in how stem cells and their progeny interface with extracellular matrix proteins and the extent to which these interactions drive specification. We have contributed to the develoment of 3D bioprinting technologies to study how content, composition and distribution of intact ECM molecules in 3D dictates specification of stem cells. Other technologies under development include multiphoton flow cytometry, an enhanced-throughput analysis and sorting modality for the study of biologic particles of hundred to thousand micron scale and delivery of stem cells to the cerebral vasculature to treat aneurysms.
We partner with departments and centers across the University of Minnesota and the international scientific community to more efficiently attain our research goals. We develop the next generation of explorers who are bold enough to merge the disciplines of biology, medicine and engineering.
Learn about our lab, get to know the people responsible for making our work possible and find out about new projects that you could join. B. Ogle